|
Planetary Energy Balance
In this lecture we discuss:
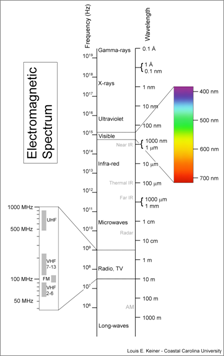
Radiation Laws All objects emit radiation. The total radiant energy is a function of the object’s temperature. The Stefan-Boltzman Law tells us exactly how much energy is emitted per unit area: E = sT4 In this equation, T is temperature (in Kelvin) and s is the Stefan-Boltzman constant (5.67e-8 Wm-2K-4). Note that E is an energy flux and has units of Wm-2. Wien’s Law tells us the wavelength of the peak radiation. It is written: lmax = b/T where b is Wien’s displacement constant (2.897e-3 mK), and T is again temperature (in Kelvin). These two laws tell us that hot objects emit lots of radiant energy at short wavelengths. We can use these two laws to determine how much energy is emitted by the Sun and the planets, and at what wavelength. The Sun, with a temperature of approximately 6000 K, emits 7.36e11 Wm-2 at a peak wavelength of 4.8e-7 m or 0.48 mm. Earth, with a temperature of only 288 K, emits 390 Wm-2 at a peak wavelength of 1.005e-5 m or 10.05 mm. The Sun and Earth emit radiation of different wavelengths (Figure 1). The Sun’s peak radiation is in the visible range; the Earth’s peak radiation is in the infra-red range.
Inverse square law All planets receive energy from the Sun. But, the amount of energy decreases with the planet’s distance from the Sun. This is because after emission, radiant energy spreads over a greater and greater area as it moves through space.
The Inverse Square Law: S = L/4pd2 is a statement of this fact, and expresses the solar flux (S) in Wm-2 as a function of solar luminosity (L), and distance from the Sun (d). Radiative equilibrium To first order, the climate of planets can be determined by radiative equilibrium. Radiative equilibrium assumes that incoming radiative energy from the Sun is equal to the outgoing radiation emitted by the planet: incoming energy = outgoing energy In other words, the planet is in energy balance. If radiative equilibrium is not achieved, then the temperature of the planet must rise or fall. We can use our radiation laws and the inverse square law to define the incoming and outgoing energy terms. Incoming energy The planets are not perfect absorbers; some of the solar radiation is reflected from the planet back to space. The reflectivity of a surface is its albedo (a). The solar radiation absorbed by the planet is then S(1-a), the solar flux minus that reflected back to space. Solar radiation is only captured by one face of a planet, the daylight side. The incident area of this side is that of a circle (pr2), where r is the planetary radius. The total incoming energy, with units W, is equal to the total absorbed flux multiplied by the incident area: S(1-a)pr2 Note that the solar flux, S, can be determined from the Inverse Square Law. Outgoing energy The Stefan-Boltzman Law determines the energy flux, or W per unit area, emitted by the planet. On average, the planet emits radiation over its entire surface, which we can assume is a sphere (4pr2). To determine the total energy emitted by the planet, we simply multiply the Stefan-Boltzman energy flux by the planetary surface area: sT44pr2 Planetary temperature Assuming radiative equilibrium, we can solve for T to get an equation for global average planetary temperature: T = (S(1-a)/(4s))1/4 If we do this calculation for Earth, using a solar flux of 1367 Wm-2 and an albedo of 0.3, we calculate a planetary temperature of 255 K. This is about 33 K less than the observed value (Table1)! Table 1. Characteristics of the terrestrial planets.
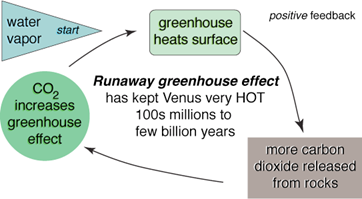
Where did our calculation go wrong? The radiative equilibrium model assumed that radiant energy from the planets is completely lost to space. In fact, both Earth and Venus have thick atmospheres that absorb some of this radiant energy and emit it back to the surface, effectively insulating the planet and raising the temperature. This process is known as the greenhouse effect, and the gases that absorb longer wave planetary radiation are known as greenhouse gases. Venus and the runaway greenhouse Venus’ surface temperature is extremely hot. Though Venus is twice as far from the Sun as Mercury (and, according to the Inverse Square Law, has one-quarter the solar flux), its surface temperature is more than 200 K higher! This is because Venus has a thick atmosphere almost completely (96%) composed of CO2, a greenhouse gas. For comparison, Earth’s atmosphere has only trace amounts of CO2. Venus’ CO2-rich atmosphere is attributed to a runaway greenhouse effect. Because Venus is relatively close to the Sun, early in its history its surface temperature was above the boiling point of water. As a result, Venus’ early oceans were evaporated, filling the atmosphere with water vapor, another greenhouse gas, and raising the planet’s temperature. As time marched on, volcanic eruptions on Venus outgassed CO2 to the atmosphere. On Earth, chemical reactions between rainwater and CO2 in the atmosphere strip CO2 out of the air and eventually lock it in sedimentary rocks. On Venus, the absence of rainfall prevented this reaction from occurring. Instead, CO2 levels climbed, and the surface temperature rose to a balmy 753 K!
Fig. 3. The Runaway greenhouse effect (http://www.astronomynotes.com/).
All materials © the Regents of the University of Michigan unless noted otherwise. |